Assembly Safety Valve Actuator
| M1114504 | |
| Neuf | |
| Soins respiratoires | |
| GE HealthCare | |
| GE HealthCare | Outright |
Enter your approval number and submit to add item(s) to cart.
Please enter approval number
OR
Don't know your approval number? Call 800-437-1171
Enter opt 1 for the first three prompts, and have your System ID available.
If you add item(s) to cart and submit your order without the
approval number, GE will contact you before your order
can be confirmed for shipment.
Select your approver's name and submit to add item(s) to your cart
Please Select Approver Name
OR
Don't know your approval number? Call 800-437-1171
Enter opt 1 for the first three prompts, and have your System ID available.
If you add item(s) to cart and submit your order without
selecting an approver, GE will contact you before your order
can be confirmed for shipment.
Features
- Part does not have evidence of visible oils, grease, dust, dirt, lint, fibers, paper, rust, chips, cleaning solutions, machining coolant, and other foreign materials on the surface
- Good dimensional stability
- Anti-static field packaging with foams for ESD sensitive materials
- Impact resistant and higher hit strength
Fréquemment acheté avec cet article
Présentation du produit
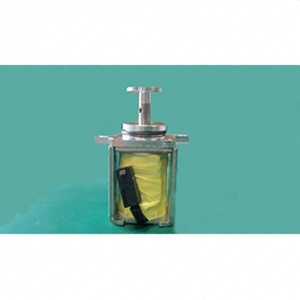
The Assembly Safety Valve Actuator is used in Respiratory Care and many other similar medical devices. It is a pull type actuator assembly having application within a ventilator inspiratory limb. An actuator assembly is designed as a pull type solenoid with an armature, spring loaded to the extended position. The armature terminates in M3 x 0.5 threads allowing choice of seal supports to be added. They are durable and compact with a good structural stability. It is made from the stainless steel material which possess excellent corrosion resistant, fire and heat resistant, high tensile strength, excellent yield strength, higher ductility, hygiene, high coefficient of friction and higher cryogenic toughness. The product is ROHS compliant and is approved for today’s safety standards. The GE product is an innovation and technology which fits well into versatile customer needs. The product is securely packaged inside a high quality packing box to avoid physical damage during transit and labeled with details about the product, Quality Assurance (QA) seal and shipment details.
Return And Exchange
GE HealthCare will accept, for return from customer, items that are in new condition, unworn, unaltered and free of damage. Items cannot be returned or exchanged beyond 30 days from customer’s receipt of the original order and all returned items will be subject to a 15% restocking fee. To obtain a full refund or exchange of the item within the 30 day return period (subject to the 15% restocking fee) customer must request a return materials authorization (RMA) from GE HealthCare and return the item to GE HealthCare utilizing this RMA number. Items must be returned in the original packaging, unused and free of damage.
Please note, cuffs, BP items, sterile and hazmat items cannot be returned. If a replacement order is requested, the restocking fee may be waived at GE HealthCare’s sole discretion.
- Mes articles récemment vus
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats
-
Se connecter pour faire des achats